
Buzzer goes off, and you have your results, which are also emailed to you.Ī friendly reminder that for all antigen tests, false positives are possible. After the step-by-step walkthrough, you swab each nostril, dip the swab in fluid, and put the fluid reader near your phone for 15 minutes. 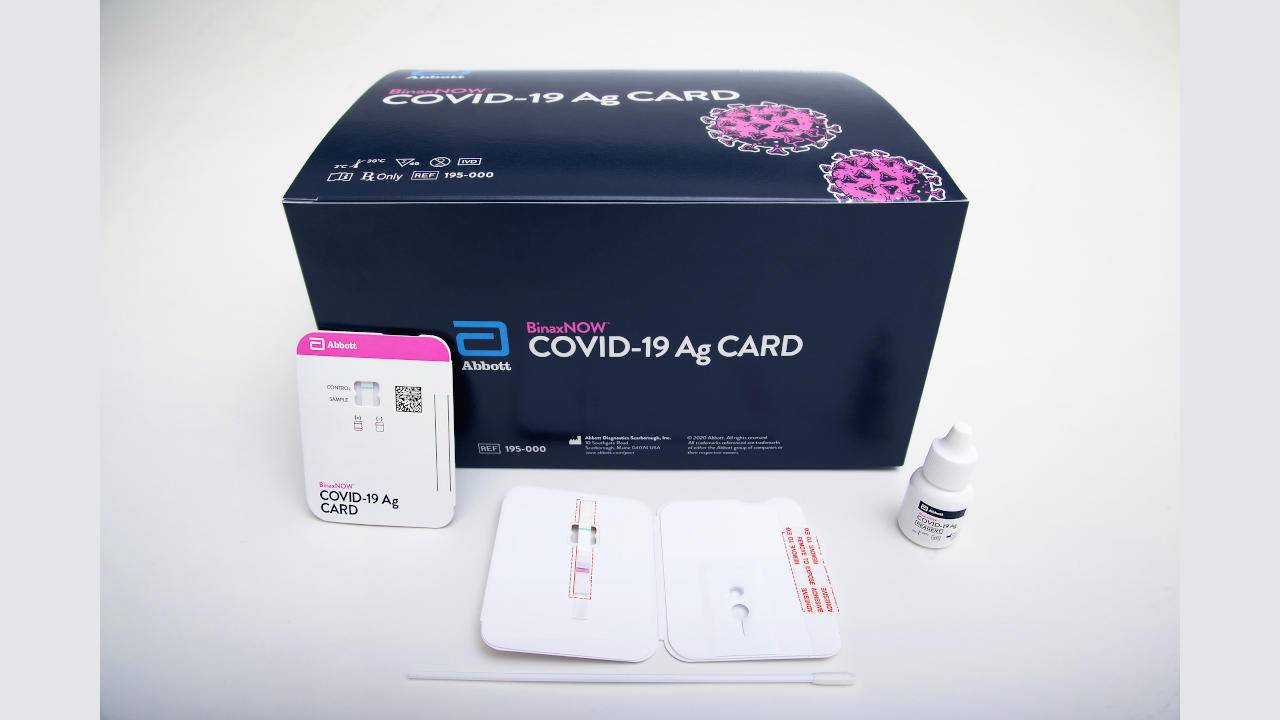
The kit has an easy-to-use smartphone app that asks you to register the bio basics of the patient (name, age, address), then it serves up a mandatory how-to video you can’t fast-forward through (a bit annoying on the fourth go, but understandably necessary). Until then, international travelers can pack one test kit for every family member over the age of two and-as long as the Wi-Fi is decent and a video-call appointment is scheduled a week out-they can take the Ellume test anywhere in the world. As a company, we’re already looking to combine our flu assets with our COVID assets to make a combined COVID-flu test, which is the natural progression. “We feel as if we’re on the cusp-we’re just about there. “The FDA knew all about our technology, from the work we’d been doing ,” Parsons told AFAR. Sean Parsons, Ellume’s CEO and founder, had been working on getting a home flu test to market since 2010 and was able to pivot-the buzzword of 2020-quickly to get approval on a COVID test. (One 15-minute test with a video observation by Azova, an online healthcare provider, will give results for both asymptomatic and symptomatic cases, for ages two and up.) How were they so speedy? Dr. Though BinaxNOW has received a bit more attention, Australia-based digital diagnostics company Ellume was actually the first to market with a COVID-19 home test, and it partnered with Alaska Airlines and Delta Air Lines to give passengers easy access to home antigen tests. “The healthcare provider comes back on camera to verify the results that you hold up to the camera, and then you can instantly see results in the app.” “They walk you through the entire process,” he says, remarking that the test is very similar to a pregnancy test-after 15 minutes or so the results pop up on the reader. His one piece of advice is to make sure to have “a solid Wi-Fi connection” so that you can download the app and conduct the telehealth video call. “It was super easy,” says Bryan Kinkade, AFAR’s publisher, who used the BinaxNOW home tests for himself and his family for a trip to Costa Rica in spring 2021. The standard kit comes with one nasal swab-the technique for sample collection is less invasive than the very deep nasal swab sample collection we’ve all heard horror stories about (if not endured ourselves). A trained telehealth professional guides users through the at-home self-test during a video call via, which will also deliver their COVID-19 test results to their email. It can be used for children as young as 4 when the sample is collected by an adult-those 15 and older can collect their own sample.īinaxNOW was developed by Abbott, a health and medical diagnostics company. It’s a rapid antigen self-test designed to detect both asymptomatic and symptomatic COVID-19. The BinaxNOW COVID-19 home test is one of the few tests on the market with FDA emergency use authorization that does not require the user to ship a sample to a lab. officials at ports of entry must be able to review and confirm your identity and paper or digital documents with your test results.įor those who would prefer to get tested in the destination, numerous airlines as well as international hotels, including in Mexico, Costa Rica, and the Caribbean, have introduced testing options and resources to help Americans fulfill the CDC testing requirement abroad.īuy now: $150 for a pack of six, The telehealth provider must issue a report confirming the patient’s identity, the name of the laboratory or healthcare entity, the type of test, and the specimen collection date.(Note that some FDA-authorized self-tests that include a telehealth service may require a prescription, according to the CDC.) The testing must include a telehealth video call during which someone authorized by the manufacturer supervises the testing procedure in real time.The test must be a SARS-CoV-2 viral test (nucleic acid amplification test or antigen test) with Emergency Use Authorization (EUA) from the U.S.To meet the CDC requirements, the tests can be self-administered as long as they meet these criteria: 
There are some potential hiccups that travelers should be aware of before they buy and pack these tests (see below for our full reviews). And as of January 15, 2022, the Biden-Harris Administration is requiring insurance companies and group health plans to cover the cost of at-home COVID-19 tests, which means they will be eligible for reimbursement through your private health coverage if you have it. Until it is, bringing a set of COVID-19 home or self-tests offers the promise of less hassle.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
